Peri-implantitis and titanium implant failure are increasingly common concerns for patients in Glen Allen, Richmond, and across Virginia. This page explores why implants sometimes fail, what symptoms to watch for, and how biological dentistry offers a whole-body, metal-free approach for long-term oral health.
Peri-implantitis is often described as a “localized” infection, but modern research and biological dentistry recognize that inflammation around a titanium implant can have whole-body implications. The mouth is a highly vascular, neurologically rich environment. When chronic inflammation develops around an implant—especially a metal implant—it does not remain isolated. Instead, a cascade of immune, inflammatory, neurological, and biochemical signals may influence systemic health.
This perspective is not about claiming titanium implants cause systemic diseases. Rather, it acknowledges that some individuals—particularly those living with autoimmune conditions, chronic inflammatory disorders, or known sensitivities—may react differently to corrosion byproducts, metal ions, and persistent oral inflammation.
Chronic oral inflammation can send inflammatory mediators—such as IL-1β, IL-6, TNF-α, and CRP—into circulation. Studies in periodontal medicine have shown clear associations between chronic oral inflammation and:
worsened blood sugar control
increased cardiovascular inflammatory markers
progression of autoimmune conditions
elevated systemic oxidative stress
chronic fatigue symptoms
flare-ups of inflammatory joint conditions
Peri-implantitis, which often produces higher bacterial loads and deeper pockets than periodontal disease, may contribute similarly to systemic inflammatory burden.
Patients in Glen Allen, Richmond, and Central Virginia often seek biological dentistry due to autoimmune diagnoses such as:
Hashimoto’s thyroiditis
Rheumatoid arthritis
Sjögren’s syndrome
Lupus
Psoriasis
Multiple sclerosis
Celiac disease
Chronic fatigue or fibromyalgia syndromes
Autoimmune conditions are known to amplify the body’s inflammatory response. When titanium corrosion products or nanoparticles enter the gum tissue or circulation, research suggests they can activate macrophages and dendritic cells—immune cells already hyperreactive in autoimmune individuals.
Asa’ad et al., 2022 showed titanium particles induce macrophages to release inflammatory cytokines.
🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC9511127/
Chen et al., 2023 documented titanium nanoparticles accumulating in peri-implant tissues, triggering inflammatory profiles.
🔗 https://www.nature.com/articles/s41368-023-00256-x
For individuals with autoimmune tendencies, these mechanisms may create a “double hit”:
existing immune dysregulation, and
local immune activation around a titanium implant.
Not every patient responds this way. But biological dentistry respects that some do.
Biological dentists also see patients with suspected or documented metal sensitivities. While metal allergy testing is imperfect, it is well established that:
titanium ions can bind with proteins
these protein–metal complexes can appear “foreign” to immune cells
sensitive patients may experience exaggerated inflammatory responses
Some patients report symptoms such as:
persistent gum inflammation
nervous system irritation
burning sensations
fatigue or brain fog (anecdotal)
inflammatory flares around implant sites
Biological dentistry does not make unsupported claims.
It simply acknowledges that individual susceptibility varies, and this variation must be respected.
The jaw is highly innervated by branches of the trigeminal nerve. Chronic inflammation—whether from bacteria, titanium corrosion, or immune activity—can irritate nearby nerve fibers.
Patients may report:
radiating pain
tingling or numbness
pressure sensations
referred symptoms to the ear, temple, or neck
burning sensations
“electric” feelings (which may be galvanic-related, see next section)
These experiences are not universal, but they are common enough among peri-implantitis cases—especially failing titanium implants—that biological dentists take them seriously.
Emerging research indicates titanium particles and ions may not stay localized.
Sharanappa et al., 2024 found elevated titanium levels in patient serum following implant placement, especially with peri-implantitis.
🔗 https://www.spandidos-publications.com/10.3892/wasj.2024.224
Toledano-Serrabona et al., 2023 discovered titanium ions and nanoparticles in the bloodstream and distant organs in an animal model.
🔗 https://aap.onlinelibrary.wiley.com/doi/epdf/10.1002/JPER.22-0091
These findings do not imply toxicity.
They simply show that titanium debris can travel systemically, which matters in patients who are immunologically sensitive.
For patients in Glen Allen, Richmond, and Virginia, understanding why titanium implants fail—and how biological dentistry interprets inflammation, corrosion, galvanism, and whole-body interaction—is essential.
Biological dentistry in Glen Allen and Richmond views peri-implantitis through a broader lens:
What is the patient’s inflammatory background?
Do they have autoimmune disease?
Are there multiple metals in the mouth?
Are galvanic currents accelerating corrosion?
Are titanium ions present in tissue or fluid samples?
Could chronic implant inflammation be affecting systemic health?
These questions do not replace conventional diagnostics—they enhance them.
They help identify patients who may benefit from closer monitoring, alternative materials, biological therapies, or earlier intervention.
Whole-body dentistry does not exaggerate the risks. It simply acknowledges that the mouth is part of the body, and inflammation anywhere matters everywhere.
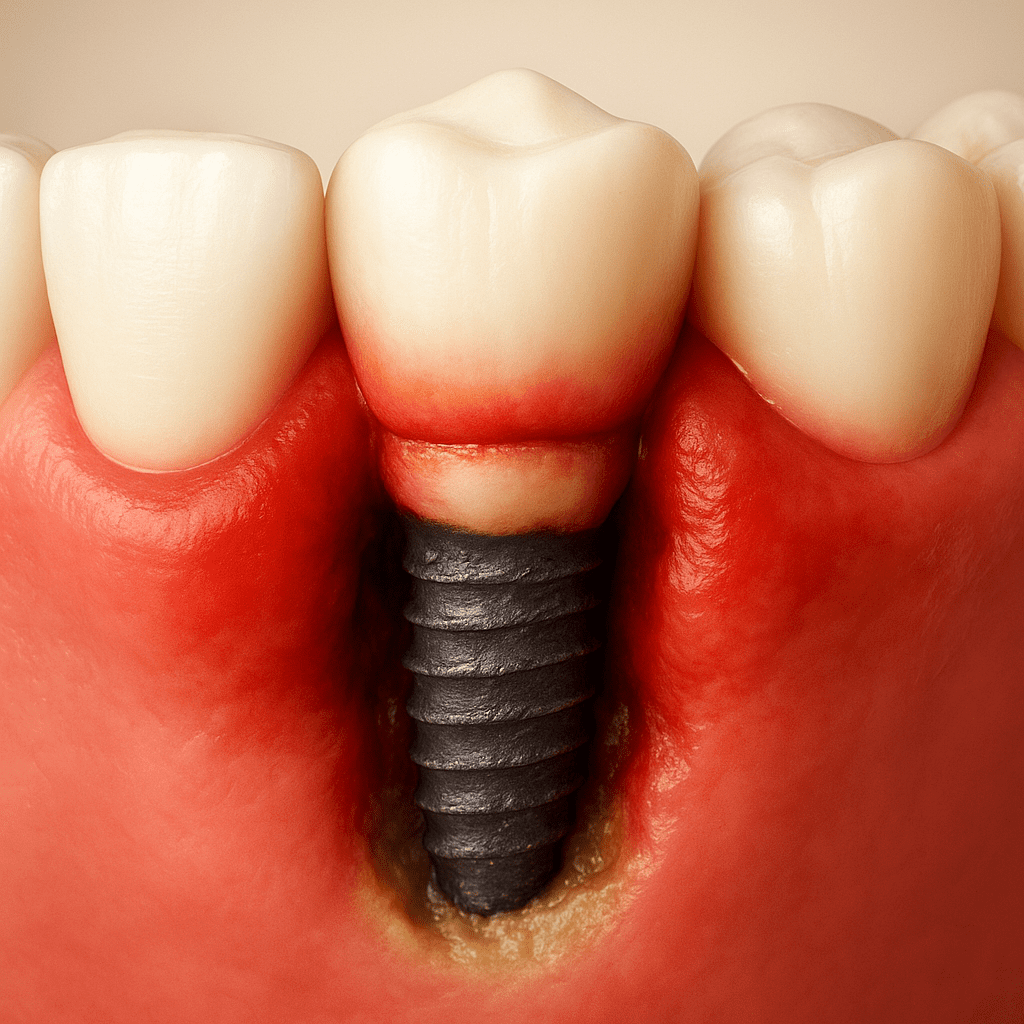
Galvanic corrosion is an important but often overlooked factor in dental titanium implant failure. It occurs when two or more different metals in the mouth—such as titanium implants, gold crowns, amalgam fillings, or stainless-steel retainers—interact through saliva, creating tiny electrical currents. Saliva acts like an electrolyte. The metals act like the two terminals of a battery. The result is micro-currents that accelerate corrosion.
Titanium implants are not isolated structures—they interact electrically with nearby metals. When galvanic currents occur, the protective oxide layer on titanium may degrade more rapidly, exposing the implant surface to:
corrosion
ion release
surface roughening
bacterial adhesion
bone loss
This “electrical stress” can make an implant more susceptible to peri-implantitis.
Kim et al. (2023) describe oral galvanism as a key factor in titanium corrosion and note that galvanic interactions can provoke mucosal irritation or sensory disturbances.
🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC10558418/
Chaturvedi (2009) identifies galvanic corrosion as one of the most common forms of corrosion affecting dental implants when multiple metals are present.
🔗 https://journals.lww.com/ijdr/fulltext/2009/20010/an_overview_of_the_corrosion_aspect_of_dental.19.aspx
Horasawa et al. (1999) demonstrated galvanic interactions between titanium and various dental alloys, showing that certain combinations significantly accelerate corrosion.
🔗 https://www.sciencedirect.com/science/article/abs/pii/S0109564199000512
MELISA Foundation (2024) provides clinician-oriented summaries comparing galvanic corrosion in different metal combinations and its potential link to inflammatory symptoms.
🔗 https://melisa.org/galvanic-corrosion/
Together, these studies reinforce the idea that mixed-metal dentistry increases the risk of corrosion and inflammatory complications around titanium implants.
Some patients experience:
metallic or bitter taste
burning mouth sensations
tingling or “electric shocks”
temperature sensitivity
unexplained irritation near the implant
faster bone loss around titanium
Not every patient will experience these, but biological clinicians are trained to consider them part of the diagnostic picture.
Patients who come to a biologically oriented practice often have:
autoimmune disorders
electromagnetic hypersensitivity
multiple metals present in the mouth
unexplained oral symptoms
For these patients, reducing galvanic load can be important.
A biological dentist may:
chart all metals in the mouth
recommend reducing polymetallic restorations when appropriate
support patients choosing metal-free implant systems
monitor symptoms related to metal sensitivity
Unlike titanium, zirconia (ceramic) implants are electrically neutral.
This means:
no galvanic currents
no corrosion
no ion release
no interaction with other metals
This is one reason patients with a history of metal sensitivities or peri-implant complications may prefer ceramic implant options—especially in biologically focused practices like those serving Glen Allen and Richmond, Virginia.
Conventional implantology generally views peri-implantitis as:
a localized bacterial infection
caused by biofilm and inadequate hygiene
progressing due to soft tissue breakdown and bone resorption
Standard explanations emphasize mechanical and microbial factors over systemic or material-related influences.
inadequate oral hygiene
history of gum disease
improper implant placement
cement trapped under the crown
lack of keratinized tissue
smoking or diabetes
too much bite force on the implant
These explanations are supported by a substantial body of research and clinical experience. For example, the 2017 World Workshop on Periodontal and Peri-Implant Diseases categorized peri-implantitis based on clinical and radiographic criteria, emphasizing inflammation and bone loss without speculation about systemic interactions.
In mainstream dentistry, peri-implantitis remains a site-specific disease, meaning:
“The problem is located at the implant, not the whole body.”
As a result, conventional treatment aims at:
cleaning the implant
reducing bacterial load
repairing damaged bone
improving hygiene techniques
sometimes surgical decontamination
However, conventional dentistry generally does not evaluate:
galvanic corrosion
metal ion release
immune hypersensitivity
systemic inflammation
whole-body contributors
These factors fall outside the traditional scope of implantology. That does not make conventional care wrong—it simply reflects its narrower clinical focus.
Biological dentistry acknowledges all the bacterial and mechanical factors described above, but adds a broader, integrative perspective. This perspective is grounded in material biocompatibility, immune individuality, and oral–systemic interactions.
Biological dentists ask deeper questions:
Why does peri-implantitis occur in some patients but not others with identical hygiene?
Why do some implants accumulate corrosion products and others do not?
Why do certain individuals experience nerve-related symptoms or burning sensations?
Could galvanic currents or metal ions contribute to the inflammatory picture?
How does the patient’s autoimmune or inflammatory background affect implant survival?
These questions are informed by a growing body of peer-reviewed research:
Titanium corrosion is well documented.
(Madi 2026)
🔗 https://pubmed.ncbi.nlm.nih.gov/41225111/
Titanium particles accumulate in peri-implant tissues.
(Chen et al. 2023)
🔗 https://www.nature.com/articles/s41368-023-00256-x
Particles activate macrophages and increase bone loss.
(Asa’ad et al. 2022)
🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC9511127/
Titanium ions appear in systemic circulation.
(Sharanappa 2024)
🔗 https://www.spandidos-publications.com/10.3892/wasj.2024.224
Galvanic currents accelerate corrosion.
(Kim 2023, Chaturvedi 2009)
🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC10558418/
🔗 https://journals.lww.com/ijdr/fulltext/2009/20010/an_overview_of_the_corrosion_aspect_of_dental.19.aspx
Using this data, biological dentistry interprets peri-implantitis as a multifactorial inflammatory condition, influenced by:
bacteria
mechanical overload
implant material chemistry
corrosion byproducts
electrical microcurrents
immune and autoimmune status
systemic inflammatory burden
Rather than treating only the site, biological dentists evaluate the whole inflammatory landscape.
Both agree on:
the diagnostic criteria (bone loss + inflammation)
the importance of early detection
the role of biofilm
the need for maintenance
the risk factors like smoking and uncontrolled diabetes
Both also agree that untreated peri-implantitis leads to implant loss.
biofilm removal
mechanical debridement
surgical access
local antimicrobial therapy
galvanic interactions
corrosion behavior
biocompatibility
systemic inflammation
autoimmune conditions
nutritional and microbiome considerations
For patients in Glen Allen and Richmond who have complex medical histories or long-standing chronic inflammation, this broader lens often feels more aligned with their lived experience.
Many patients who seek a biological dentist already arrive with:
longstanding implant inflammation
symptoms not explained by conventional models
concerns about metal sensitivity
autoimmune or chronic inflammatory conditions
the desire for metal-free options
Biological dentistry offers an expanded framework grounded in both science and whole-body awareness.
When a titanium implant fails due to bone loss, corrosion-related inflammation, or patient sensitivity, many individuals look toward ceramic (zirconia) implants, particularly SDS Swiss Dental Solutions implants, because they offer a fully metal-free approach with high biocompatibility.
Below is a research-supported, patient-friendly overview of why ceramic implants are increasingly preferred in biological dentistry.
Zirconia is a stabilized ceramic material—not a metal and not conductive.
It does not corrode, produce ions, or participate in galvanic reactions.
Benefits:
zero metal ion release
no electrical currents
no galvanic pain or metallic taste
no risk of metal hypersensitivity
lower bacterial adhesion compared with titanium
This matters especially for individuals with:
autoimmune disease
chronic inflammation
metal sensitivity
past reactions to metals
prior titanium implant failure
Several studies indicate that zirconia surfaces accumulate fewer early bacterial colonies compared with titanium.
For example:
Chiou et al., 2023 found reduced early-stage biofilm on zirconia compared with titanium.
🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC10122594/
Kniha et al., 2021 observed different bacterial colonization patterns favoring zirconia.
🔗 https://www.sciencedirect.com/science/article/abs/pii/S0901502721000400
Because peri-implantitis is fundamentally a biofilm-driven disease, a surface that attracts less bacteria is a major advantage.
Soft tissues around zirconia implants behave more like natural gums.
tighter epithelial attachment
reduced inflammation
better blood supply
improved mucosal stability
Abouel Maaty et al., 2025 found that zirconia abutments led to more favorable inflammatory profiles compared with titanium.
🔗 https://www.mdpi.com/2073-4409/14/2/129
In a biological practice, this is significant because healthier soft tissue reduces bacterial penetration and lowers peri-implantitis risk.
Zirconia is chemically inert.
It does not rust, oxidize, or leach ions.
This eliminates the entire category of titanium-associated concerns:
corrosion
particle debris
titanium serum elevation
macrophage-driven cytokine storms
bone loss caused by corrosion products
Ceramic implants remove these variables entirely.
Because zirconia does not shed ions or nanoparticles, several studies suggest:
lower macrophage activation
reduced inflammatory cytokines
stable bone levels
Hanawa (2020) emphasized zirconia’s excellent biocompatibility and stable tissue integration.
🔗 https://www.jstage.jst.go.jp/article/dmj/39/1/39_2019-172/_pdf
Modern zirconia implants—especially SDS designs—demonstrate excellent survival.
Mohseni et al., 2023
Zirconia implants show survival rates comparable to titanium.
🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC10746607/
Padhye et al., 2023
Reviews conclude ceramic implants perform similarly to titanium clinically.
🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC10630218/
Gul et al., 2024
Reported ~95% short-term survival with good stability.
🔗 https://www.jdentistry.com/article/S0300571224000733
As SDS expands clinical trials (NCT06314425), ceramic outcomes continue to strengthen.
Zirconia is naturally white.
It avoids the gray shine-through that titanium can produce, especially in patients with thin gums.
SDS ceramic implants also support:
one-piece monoblock design
reduced microgaps
minimized bacterial infiltration
excellent biomimetic crown emergence
Patients seeking biological dentistry often choose SDS implants because:
they want a metal-free solution
they have experienced titanium-related symptoms
they have autoimmune or inflammatory conditions
they reacted poorly to previous implants or metals
they prefer a practice that considers whole-body health
For these reasons, SDS ceramic implants align extremely well with biocompatibility-focused, holistic dental care for patients in Glen Allen and Richmond, Virginia.
Thinking about implant health?
If you live in Glen Allen, Richmond or anywhere in Virginia and are concerned about titanium implants, metal sensitivity, or long-term oral health — call or click to schedule a biological implant health consultation.
We assess your full-body history, oral environment, and implant status to help you make an informed choice.
Click here to make an appointment now or call (804) 381-6238 or email at info@virginiabiologicaldentistry.com to learn more.

Biological dentistry is not opposed to titanium implants, nor does it assume they must be removed. Many patients do well with titanium for decades.
However, some individuals develop complications—including progressive bone loss, chronic inflammation, corrosion-related symptoms, or confirmed metal sensitivities. These patients often seek biological dentists because they want:
a metal-free alternative,
a more biocompatible approach, or
guidance after documented peri-implantitis or implant failure.
An implant removal is not recommended unless there is clear dental and health justification, such as:
progressive or irreversible bone loss
implant mobility
persistent infection or suppuration
mechanical or structural fracture
functional impairment affecting chewing or comfort
In these situations, removal becomes a clinical decision, not a philosophical one. Patients who elect ceramic implant replacement typically do so for:
medical or immunological concerns,
a desire for a fully metal-free solution,
previous sensitivity reactions,
biologically focused long-term wellness, or
aesthetic preferences.
The role of a biological dentist is to provide an objective, evidence-based evaluation, discuss all appropriate options, and support the patient in choosing the path that aligns with their oral health needs, medical history, and personal values—without judgment, pressure, or bias.